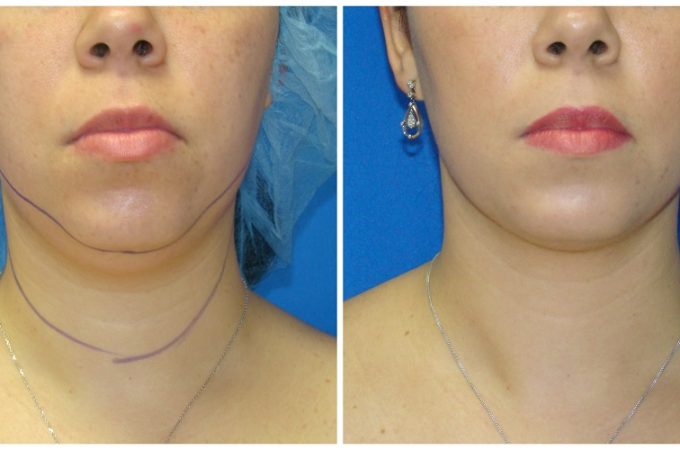
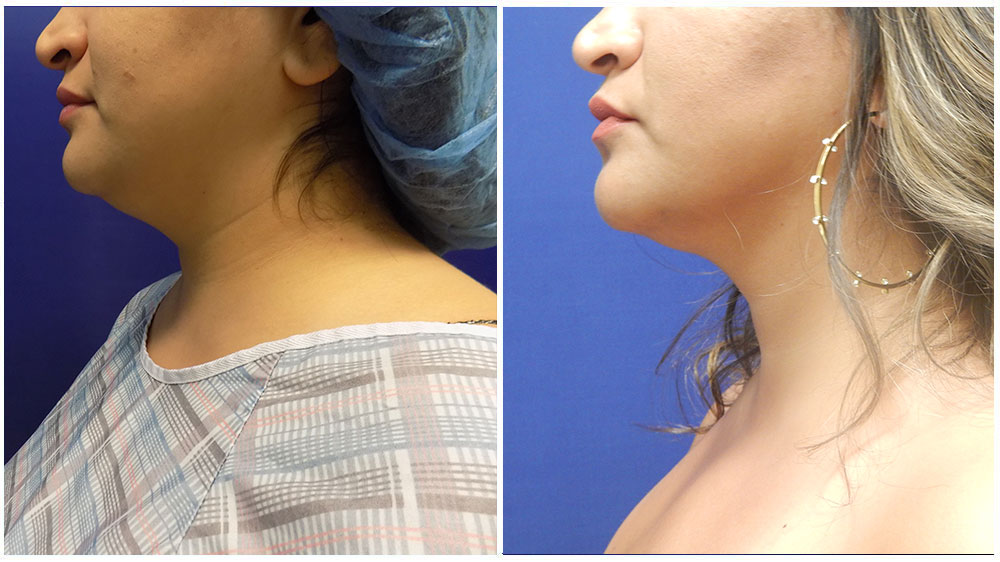
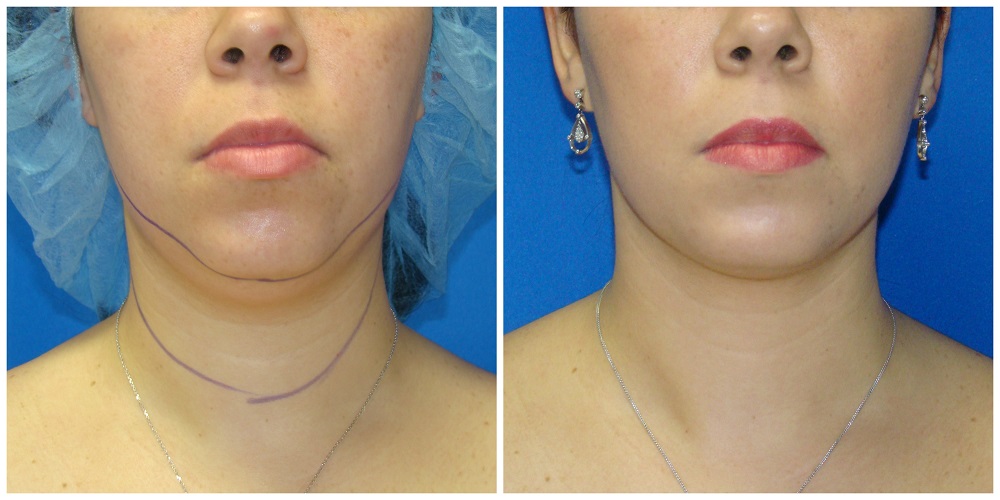
Is liposuction for love handles safe? Absolutely. Liposuction is a safe and effective way to remove stubborn fat pockets from the body’s midsection. Recovering from love-handle liposuction can take some time, but with proper aftercare, there are ways to speed up healing while ensuring best results. Here’s what you should know about recovery and how to get back on your feet as quickly as possible.
The Recovery Process
1: Preparing for Surgery
Prior to undergoing any surgery, it is important to prepare both physically and mentally. Make sure that you have someone available to drive you home afterward and do light chores around the house for the first few days. It is also important to talk with your doctor about any medications or supplements that may need to be stopped before going into surgery. Finally, arrange for plenty of rest in the days leading up so that your body is well-rested and ready for healing after the procedure.
2: What To Expect After Surgery
After surgery, expect some discomfort as your body adjusts to its new shape. Immediately following the procedure, patients often experience pain, tenderness, swelling around the treatment area, bruising, minor bleeding and temporary numbness or tingling sensations which normally subside within a week or two following surgery. Most patients also find they cannot stand up immediately; this will improve once swelling reduces and your core muscles regain their strength during recovery.
3: How Long Does Swelling Last?
Swelling can last anywhere from a week to several months, depending on individual factors such as age, pre-surgery activity level and post-surgery lifestyle choices such as diet quality and exercise patterns. During this time, it’s best to avoid strenuous activities such as running or heavy lifting, as this could lead to further complications if we don’t allow our bodies to heal naturally at their own pace without forcing them too soon.
4: Compression garments
Wearing compression garments immediately after surgery helps reduce inflammation by supporting the natural healing. It also helps to minimize swelling in all areas of treatment while allowing lymphatic drainage, which leads to faster recovery times. These garments are usually worn during the day, but can be taken off at night when sleeping or relaxing at home if required.
5: Diet and exercise after treatment
During recovery, it is highly recommended that you do nothing too strenuous and follow a healthy diet with plenty of fresh fruit, vegetables, lean protein, complex carbohydrates (such as whole grains) and plenty of water (8 glasses a day). Once these basic dietary guidelines have been met, gentle exercise such as walking, swimming, yoga etc… can be started slowly under the supervision of a healthcare professional if required.
6: Follow-up appointments with your surgeon
It is always very important to keep scheduled follow-up appointments with your surgeon or medical team so that they can monitor the progress made during the recovery period, especially if there have been any unforeseen circumstances in the early stages after treatment. This will allow us to make any necessary adjustments to ensure the best overall outcome.